Ageing is associated with an impaired cellular function that can affect tissue architecture and wound healing in gingival and periodontal tissues, However, the impact of oral fibroblast ageing on the structural organization of the extracellular matrix (ECM) proteins is poorly understood. In this study, the impact of cellular ageing of oral fibroblast is investigated on the production and structural organization of collagen and other ECM proteins.
Oral fibroblasts were serially subcultured, and replicative cellular senescence was assessed using population doubling time, Ki67 counts and expression of P21WAFI. The production and structural organization of ECM proteins were assessed at an early (young-oFB) and late (aged-oFB) passages. The thickness and pattern of collagen produced by live cultures of young- and aged-oFB were assessed using a label-free and non-invasive second harmonic generation (SHG)-based multiphoton imaging. Expression of other ECM proteins (fibronectin, fibrillin, collagen-IV and laminins) were evaluated using immunocytochemistry and confocal microscopy-based depth profile analysis.

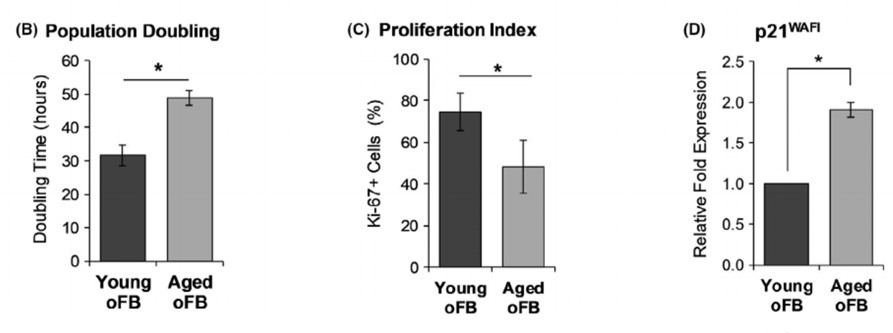 A) Effect of aging on cell morphology, proliferation and expression of senescence-associated P21WAFI. B) Photomicrographs display the morphological difference under phase contrast microscopy and expression of Ki67 among of young and aged phenotypes of oral fibroblasts. The graphs display the replicative senescent phenotype of aged-oFB as represented by higher population doubling time. C) Lower percentage of Ki67+ cells and D) Higher expression of senescence-associated P21WAFI.
A) Effect of aging on cell morphology, proliferation and expression of senescence-associated P21WAFI. B) Photomicrographs display the morphological difference under phase contrast microscopy and expression of Ki67 among of young and aged phenotypes of oral fibroblasts. The graphs display the replicative senescent phenotype of aged-oFB as represented by higher population doubling time. C) Lower percentage of Ki67+ cells and D) Higher expression of senescence-associated P21WAFI.
Aged-oFB displayed a higher population doubling time, lower Ki67+ cells and higher expression of P21WAFI indicative of slower proliferation rate and senescence phenotype. SHG imaging demonstrated that young-oFB produced a thick, interwoven network of collagen fibres, while the aged-oFB produced thin and linearly organized collagen fibres. Similarly, analysis of immunostained cultures showed that young-oFB produced a rich, interwoven mesh of fibronectin, fibrillin and collagen-IV fibres. Lastly, there was no observable difference in production and organization of laminins among the young- and aged-oFB.
The results suggest that oral fibroblast ageing impairs ECM production and more importantly the organization of ECM fibres, which could potentially impair wound healing in the elderly.
The full article can be accessed here.
1Faculty of Dentistry, National University of Singapore, Singapore City, Singapore, 2A*STAR Microscopy Platform, Research Support Centre, Agency for Science, Technology and Research (A*STAR), Singapore City, Singapore, 3Bioinformatics Institute, Agency for Science, Technology and Research (A*STAR), Singapore City, Singapore